MTPConnect is a not-for-profit organisation, established by the Australian Federal Government’s Industry Growth Initiative. MTPConnect’s aim is to accelerate the rate of growth of the medical technologies, biotechnologies and pharmaceuticals (MTP) sector to increase commercialisation, collaboration and establish Australia as an Asia-Pacific hub...
SeerPharma
Recent Posts
SeerPharma is excited to be sponsoring BioMelbourne Network’s BioSymposium focusing on Devices + Diagnostics – Navigating our New World. The event to be held Thursday the 29th of April.
Let’s just say what we’re all thinking: 2020 was rough. It threw every element of our lives into disarray. We suddenly had to become experts at navigating a new way of working, schooling and socialising. It was disruptive and it was challenging, but it was also transformative. And while the past 12 months might have felt like 12 years, it’s time...
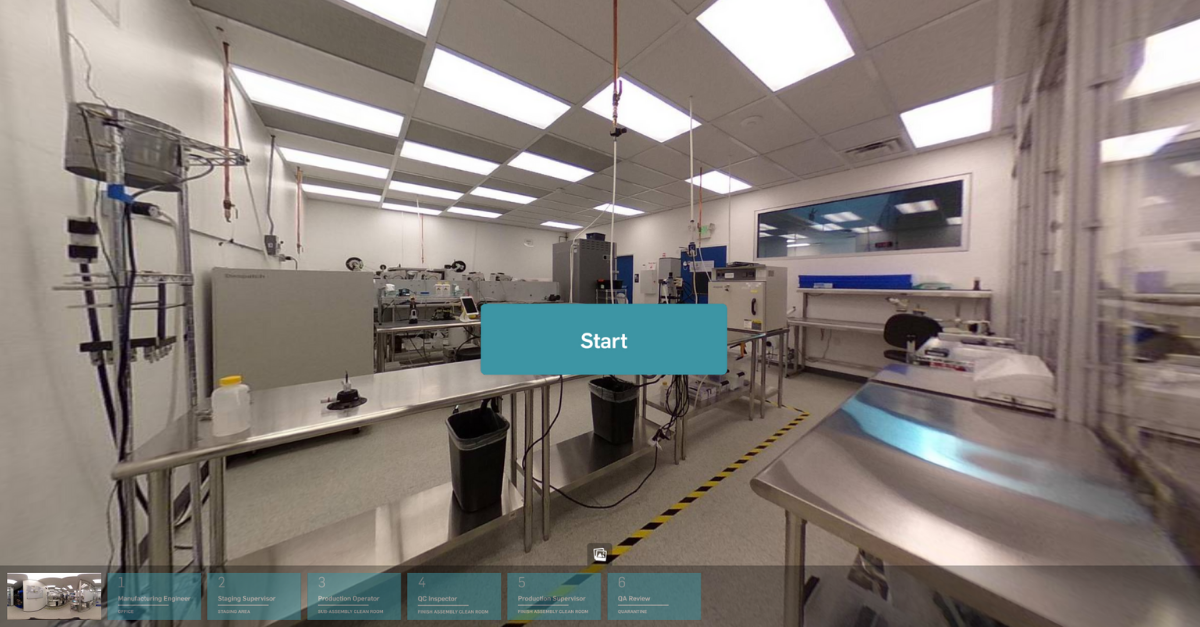
MasterControl Manufacturing Software Solutions
MasterControl delivers the next level of manufacturing excellence by bridging the gap between performance and quality with digital production records that finally let you go 100% paperless.
MasterControl Manufacturing Excellence (Mx)
Shaping the Next Normal for Quality and Compliance: 2021 QM Trends
Let’s just say what we’re all thinking: 2020 was rough. It threw every element of our lives into disarray. We suddenly had to become experts at navigating a new way of working, schooling and socialising. It was disruptive and it was challenging, but it was also transformative. And although the past 12 months might have felt like 12 years, it’s...
iNova Pharmaceuticals Engage SeerPharma to Support CSV Operating Model
iNova Pharmaceuticals (“iNova”) is an international organisation that develops, markets and sells a wide-range of prescription medicines and non-prescription consumer health products to over 20 countries across Asia, Australia and New Zealand, and Africa. Employing over 500 industry professionals over 3 continents, iNova’s vision is to deliver...
Adapting to Pharma’s Next Normal: 2021 Pharmaceutical Trends
Let’s just say what we’re all thinking: 2020 was rough. It threw every element of our lives into disarray. We suddenly had to become experts at navigating a new way of working, schooling and socialising. It was disruptive and it was challenging, but it was also transformative. And although the past 12 months might have felt like 12 years, it’s...
Insights into the World of Extractables and Leachables
Overview
An understanding of factors that can impact product purity, is a crucial component of assuring the quality of pharmaceuticals released to market. Regulatory bodies such as the US FDA and the European Medicines Agency (EMA) are increasingly focusing on the interactions between the various steps involved in manufacturing the final...
Apply Now to Start Online GMP Postgraduate Studies in February 2021
SeerPharma has partnered with the Graduate School of Health at the University of Technology Sydney (UTS) to deliver postgraduate qualifications in Good Manufacturing Practice (GMP). SeerPharma and UTS have worked tirelessly in the background to migrate this course for online delivery over the last few years to provide working professionals the...